We study some of the molecular mechanisms that allow bacterial cells to grow and divide so efficiently. Our main model organism is the Gram+ bacterium Bacillus subtilis and we apply a broad combination of approaches, from structural biology to live cell microscopy. We focus on fundamental questions but also apply our discoveries and expertise to validate new antibiotic targets.
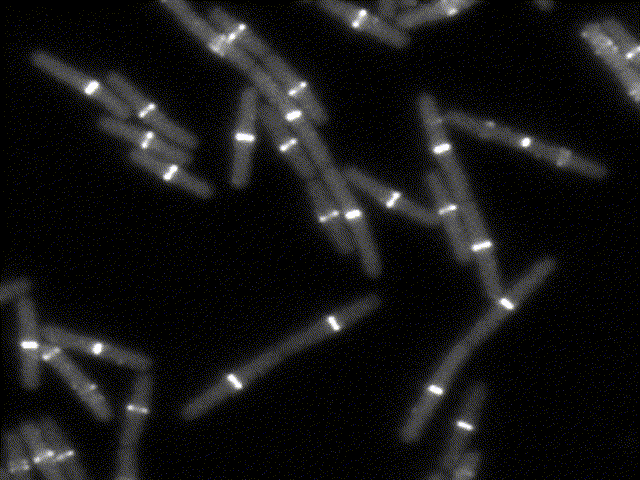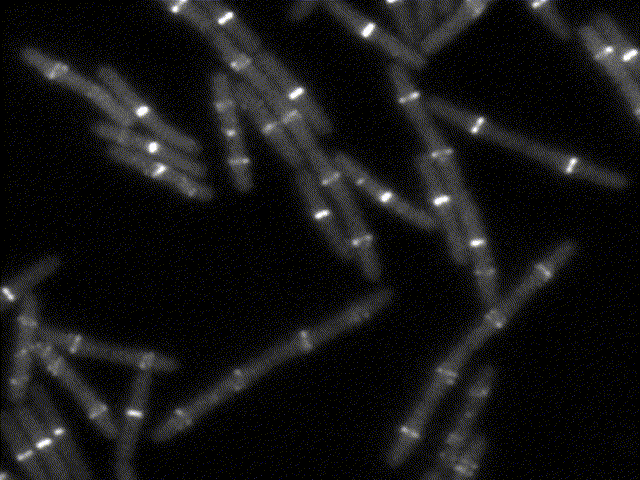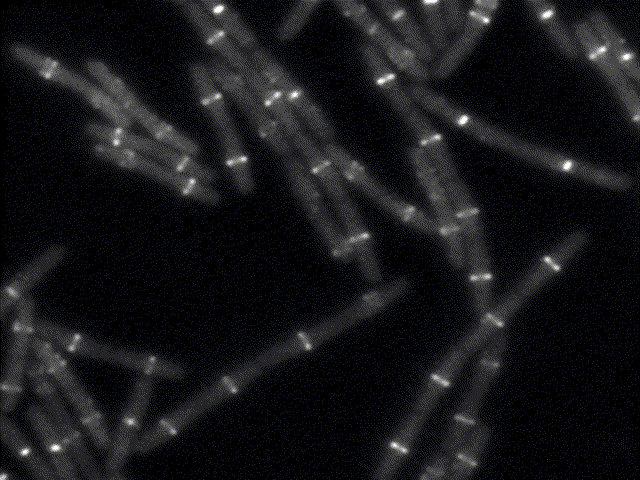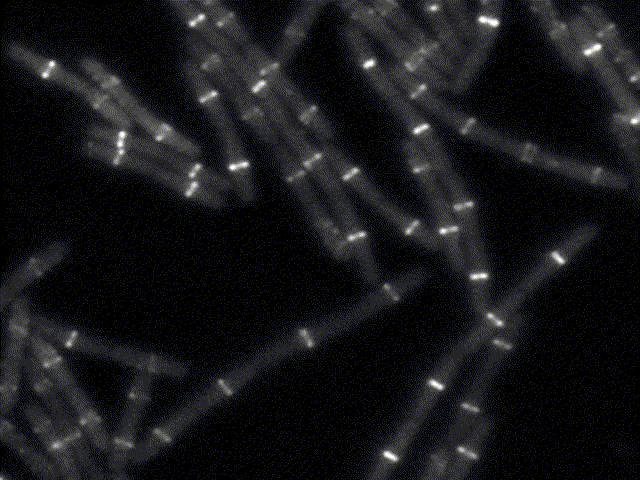
How bacteria know where to divide?
This movie shows the bacterial division apparatus in action (beware: it´s been sped up about 100X !). It is a remarkable protein machine controlled by FtsZ, the bacterial homolog of tubulin. FtsZ sets the location and timing of division by self-assembling into a cytoskeletal structure - the Z ring - which serves as tracks to guide the inward synthesis of cell wall to produce the division septum. We study the proteins that regulate Z-ring assembly to ensure that division happens only at midcell and in perfect coordination with DNA replication. We apply genetics, biochemistry and structural biology to understand how such regulatory proteins affect the assembly of FtsZ and how this explains the spatio-temporal control of cell division. Because some of these regulatory proteins (MinC, MciZ) are inhibitors of FtsZ function, we hope that deciphering of their mechanisms will generate insight for the design of cell division inhibitors with therapeutic potential.
How bacteria know when to grow and when to stop?
In contrast to what is commonly thought, bacterial growth does not cease because of lack of nutrients. It has to cease before cells run out of nutrients, to avoid the damages of error-prone and unbalanced biosynthetic processes (you don't want to keep running an engine without coolant in the radiator, isn't it?). A central molecule in informing the cell about nutrient availability is the nucleotide second messenger (p)ppGpp, also known as magic spot or alarmone. Normally present at a low basal level, the concentration of this nucleotide increases sharply when cells face starvation of a variety of nutrients, and also under certain stresses. Accumulation of (p)ppGpp then shuts down biosynthesis and redirects resources for survival functions instead of growth. We have been studying this signalling pathway and its importance when cells are starved for lipids. We have shown that (p)ppGpp is essential for surviving inhibition of fatty acid synthesis, and that cells that lack this signalling nucleotide do not notice that they are starved for lipids and kill themselves by continuing to grow in the absence of new membrane synthesis (very much like the car analogy above). This uncovered a very important role of (p)ppGpp in keeping biosynthetic processes coordinated and the dangers that ensue when coordination is lost. Because cells become highly vulnerable when they lack (p)ppGpp we are exploring this pathway as a novel antibiotic target. A simple video describing this project can be found here.


How bacteria make their membranes?
We still do not understand how the plasma membrane of bacteria is made. The enzymes and chemical reactions that produce phospholipids have been worked out, but how the insertion of new building blocks is organized in space and time, and how membrane synthesis is coordinated with cell growth remain fundamental questions of bacterial cell biology. There is still controversy whether membrane synthesis is restricted to specific locations, such as the division site, or happens throughout the cell, and to solve this we are applying modern fluorescence microscopy methods to determine the subcellular localization and dynamics of phospholipid-synthesizing enzymes and whether they function alone or in a multiprotein complex. We are also trying to devise the mechanisms that keep membrane synthesis coordinated with other biosynthetic processes. We have shown that part of the answer is ppGpp, since in the absence of this signal growth continues even when membrane synthesis is inhibited, leading to catastrophic death. We are still trying to understand how bacteria avoid making more membrane than necessary to encase their volume, something that is likely controlled by the acyl-transferase PlsX, the first enzyme in the path for phospholipid synthesis.
